Neuroscientists have discovered that a familiar enzyme has a secret, powerful ability to protect brain cells, opening new doors for the fight against neurodegenerative diseases like Alzheimer’s.
In the intricate landscape of the human brain, a constant battle is waged at the cellular level. The enemy is oxidative stress—a destructive imbalance that occurs when harmful molecules called oxidants overwhelm the brain’s natural antioxidant defenses. This relentless assault is a known hallmark of aging and a key driver of devastating neurodegenerative diseases, including Alzheimer’s. For decades, scientists have searched for ways to bolster the brain’s defenses. Now, a groundbreaking study from Johns Hopkins Medicine has revealed a powerful, previously unknown protective mechanism, centered on an enzyme we thought we already understood.
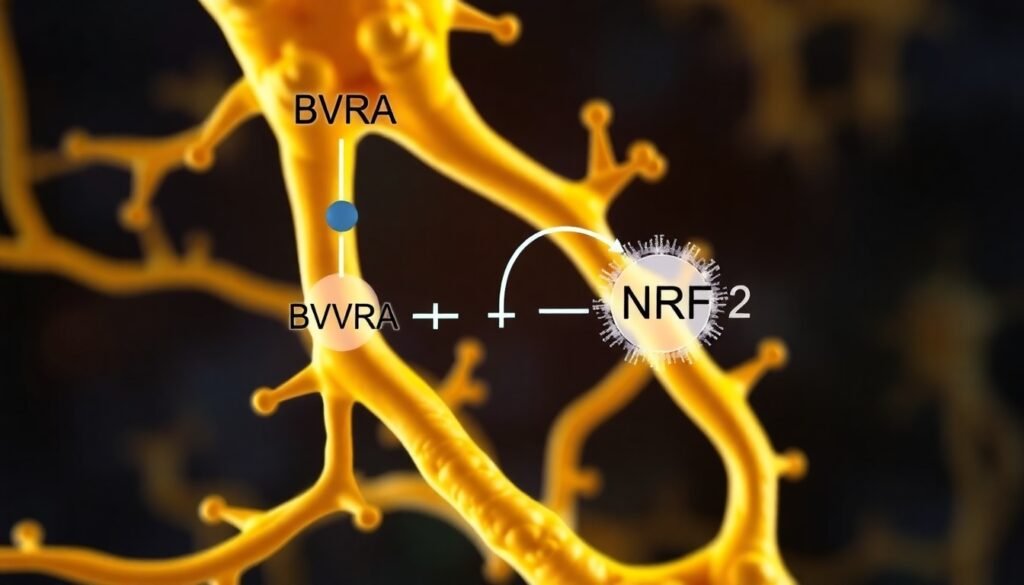
New research shows that an enzyme named biliverdin reductase A (BVRA) acts as a direct and crucial guardian of our neurons, and it does so in a way that completely changes our understanding of its function. This discovery not only rewrites a chapter in our neurobiology textbooks but also provides a promising new target for therapies aimed at preserving brain health and cognition.

More Than Just a Pigment Producer
Until now, BVRA was primarily known for one specific job: producing bilirubin. Bilirubin, the pigment responsible for the yellow color of bruises and jaundice, is the final product of heme catabolism (the breakdown of the oxygen-carrying component of blood). For years, scientists have recognized that bilirubin itself is a potent antioxidant, capable of neutralizing damaging oxidants in the brain. Because of this, it was naturally assumed that BVRA’s protective benefits were entirely due to its role as a bilirubin factory. The more bilirubin it made, the more protection the brain received. It was a simple and logical conclusion.
The recent findings, published in the Proceedings of the National Academy of Sciences, have turned this assumption on its head. A team of researchers led by Dr. Bindu Paul, an associate professor at the Johns Hopkins University School of Medicine, decided to investigate if there was more to BVRA’s story. Their work revealed that BVRA has a vital, non-enzymatic role that is completely independent of its ability to produce bilirubin.
A Master Switch for Cellular Defense
The secret to BVRA’s newfound power lies in its direct interaction with another critical protein: nuclear factor erythroid-derived factor-like 2, or NRF2. Think of NRF2 as a master switch for the cell’s entire antioxidant defense system. When activated, NRF2 travels to the cell’s nucleus and turns on a suite of genes responsible for producing protective proteins and antioxidants. This coordinated response is essential for maintaining cellular health and resilience against stress.
The Johns Hopkins team discovered that BVRA physically binds to NRF2. This binding appears to be the key that unlocks NRF2’s full potential, ensuring the antioxidant defense system is robust and responsive. Without BVRA’s direct involvement, the NRF2 system falters, leaving neurons vulnerable to damage.
"Our research identifies BVRA as a key player in cellular defense with profound implications for aging, cognition and neurodegeneration," explains Dr. Paul. This interaction positions BVRA not just as a simple enzyme, but as what first author Dr. Chirag Vasavda calls a "molecular integrator of key cellular processes that help protect neurons from damage."
The Evidence from the Lab
To prove this novel connection, the scientists conducted a series of elegant experiments using genetically engineered mice. Their first clue came from an experiment where they tried to create mice that lacked the genes for both BVRA and NRF2. None of the mice survived, a stark indication that the two proteins have a deeply intertwined and essential function.
Next, they examined mice engineered to lack only the BVRA enzyme. In these mice, the researchers observed that the NRF2 protein malfunctioned. Its ability to activate its target genes was impaired, resulting in significantly lower levels of protective antioxidants in the brain cells. This demonstrated that NRF2 needs BVRA to do its job properly.
The most compelling piece of evidence came from a final experiment. The team created a mutant version of the BVRA enzyme that was incapable of producing bilirubin. Its primary, long-understood function was completely disabled. When this mutant BVRA was introduced into neurons, it still retained its ability to bind to NRF2 and protect the cells from oxidative stress. This was the smoking gun: BVRA’s role as a guardian of the brain is not just about making bilirubin; it has a second, equally important job as a direct regulator of the NRF2 defense pathway.
A New Hope for Treating Neurodegeneration
This discovery has significant therapeutic potential. Oxidative stress is a major contributor to the neuronal death seen in Alzheimer’s disease and other neurodegenerative conditions. For a long time, the therapeutic approach has focused on supplementing the body with external antioxidants, with limited success. This new research suggests a more sophisticated strategy: instead of just adding more antioxidants, what if we could empower the brain to ramp up its own, highly efficient defense systems?
By targeting the newly discovered BVRA-NRF2 axis, it may be possible to develop drugs that strengthen this natural protective pathway. "This role of BVRA could potentially be targeted by drugs to slow the development of neurodegenerative disorders such as Alzheimer’s disease,” says co-corresponding author Dr. Solomon H. Snyder, a distinguished professor at Johns Hopkins.
The research team’s next step is to investigate how this BVRA-NRF2 connection goes wrong in mouse models of Alzheimer’s disease. Understanding how this pathway fails could provide the final clues needed to design effective interventions. This work, the result of a years-long, multidisciplinary effort, highlights how investing in fundamental scientific discovery can pave the way for the medical breakthroughs of tomorrow, offering a new beacon of hope in the ongoing battle to protect our most vital organ.
Reference
Paul, B., Vasavda, C., Kothari, R., Chakraborty, S., Tripathi, S. J., Shanmukha, S., … & Snyder, S. H. (2024). Biliverdin reductase A is a major determinant of protective NRF2 signaling. Proceedings of the National Academy of Sciences, 121(41), e2408211121.