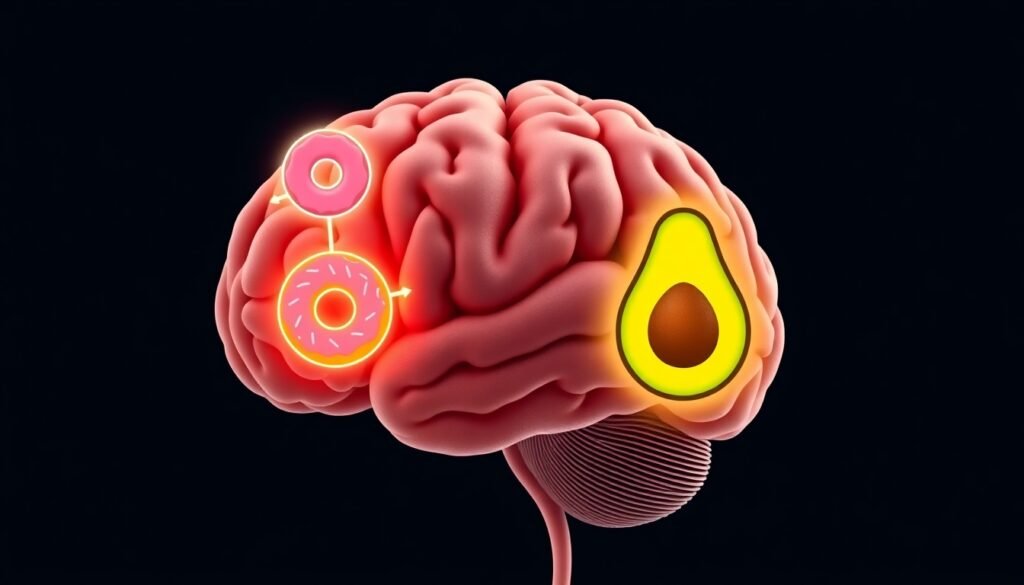
New research reveals that when your energy dips, your brain doesn’t just scream ‘eat!’—it sends out two distinct orders, one for quick-energy carbs and another for long-lasting fats.
It’s 3 PM. The familiar afternoon slump hits, and with it, a powerful craving. But what do you reach for? Is it the sugary donut calling your name, promising a quick jolt of energy? Or is it the savory, fatty crunch of potato chips that seems irresistible? We often treat cravings as a monolithic force, a simple demand for "food." However, the specificity of these desires—the sudden, intense need for sugar versus fat—points to a much more sophisticated system at play within our brains. For years, scientists have worked to map the neural circuits that control hunger, but a central question remained elusive: how does the brain distinguish between different types of nutrients when it sends out its signals? A groundbreaking new study in mice has finally shed light on this mystery, revealing that the brain uses two completely separate neural highways to regulate our intake of carbohydrates and fats.
The Body’s Energy Crisis: The Starting Gun for Cravings
To understand how these circuits work, we first need to understand the trigger. Imagine your body’s cells are running on a fuel tank filled with glucose (sugar). When the fuel level drops—a state scientists call glucoprivation—an alarm sounds throughout your nervous system. This isn’t just a feeling of low energy; it’s a critical biological signal that the body needs to refuel, and fast. This energy deficit is the starting gun that sets the brain’s complex food-seeking machinery in motion. Researchers in a recent study simulated this state in mice by administering a compound called 2-deoxy-d-glucose (2DG), which effectively tricks the body into thinking its blood sugar has plummeted. This allowed them to observe precisely what happens in the brain when it faces an energy crisis.
Inside the Command Center: Two Distinct Neural Pathways
The brain’s command center for basic needs like hunger, thirst, and body temperature is a small but powerful region called the hypothalamus. Within this hub, a specific area known as the paraventricular nucleus (PVH) acts as a switchboard, directing the body’s response to the energy alarm. The new research discovered that this switchboard operates two distinct and independent lines of communication, one for carbohydrates and one for fats.
These communications are initiated by specialized messenger cells called neuropeptide Y (NPY) neurons. These neurons are the first responders; they detect the drop in glucose and carry the "we need energy" message to the PVH. But here’s where it gets fascinating: different groups of NPY neurons deliver different instructions.
Highway 1: The Express Lane for Carbohydrates
When glucoprivation occurs, NPY neurons located in the brainstem (specifically, the nucleus of the solitary tract and ventrolateral medulla) are activated. They send a signal directly to the PVH, targeting a group of cells known as corticotropin-releasing hormone (CRH) neurons. This activation, regulated by an energy-sensing enzyme called AMP kinase (AMPK), is essentially a "Code Red" for sugar. It drives an immediate and powerful craving for high-carbohydrate foods. This makes perfect evolutionary sense. When energy is critically low, the fastest way to replenish it is by consuming simple sugars. This pathway is the brain’s quick-fix mechanism, designed to rapidly restore blood glucose levels and resolve the immediate crisis. In the study, when this circuit was active, the mice preferentially sought out the high-carbohydrate diet (HCD).

Highway 2: The Stock-Up Signal for Fats
At the same time, a second, parallel process unfolds. NPY neurons—both from the brainstem and another part of the hypothalamus called the arcuate nucleus (ARC)—also send signals to the PVH. However, they target a different set of cells: the melanocortin 4 receptor (MC4R) neurons. Instead of activating them, the NPY neurons inhibit them.
This might seem counterintuitive, but think of the MC4R neurons as gatekeepers that normally put the brakes on fat consumption. By inhibiting these gatekeepers, the NPY signal effectively lifts the restrictions on eating high-fat foods. This is not about a quick energy fix; it’s about long-term strategy. Fats are incredibly energy-dense, making them the perfect fuel to store for future needs. This circuit tells the body, "The immediate crisis is being handled, now let’s stock up the reserves to make sure this doesn’t happen again soon." The study confirmed this elegant mechanism: inhibiting the MC4R neurons led the mice to consume significantly more of the high-fat diet (HFD). This reveals that the brain has a dual strategy: solve the immediate problem with carbs while planning for the future with fats.
Why This Discovery Matters for Human Health
While this research was conducted in mice, the neural pathways governing hunger are remarkably conserved across mammals, including humans. This discovery opens up a new frontier for understanding and treating a range of metabolic and eating disorders.
For decades, the approach to treating obesity has often focused on general appetite suppression, which can have broad and sometimes undesirable side effects. This new understanding of separate nutrient-specific circuits could pave the way for highly targeted therapies. Imagine a treatment that could specifically dampen the circuit driving cravings for unhealthy fats or sugars without affecting the overall feeling of hunger or the desire for nutritious foods.
Furthermore, this research provides critical insights into conditions like binge eating disorder, where cravings can become overpowering and dysregulated. By pinpointing the exact neural players involved in specific food choices, we may be able to develop interventions that restore balance to these circuits. It also helps explain why, under stress or when energy is low, we might find ourselves battling intense, specific cravings that feel impossible to ignore. It’s not a lack of willpower; it’s a finely tuned biological system executing a specific command.
A More Nuanced View of Hunger
The next time you feel a specific craving, remember that your brain is running a complex diagnostic program. It’s not just sending a generic "feed me" signal; it’s making a calculated decision based on your body’s immediate and long-term energy needs. This research beautifully illustrates that our relationship with food is governed by an incredibly sophisticated and nuanced neural architecture. By untangling these separate pathways for sugar and fat, scientists have not only solved a long-standing puzzle in neuroscience but have also provided a new roadmap for developing smarter, more effective strategies to promote human health.
Reference
Rattanajearakul, N., et al. (2024). Glucoprivation-induced nutrient preference relies on distinct NPY neurons that project to the paraventricular nucleus of the hypothalamus. Metabolism.